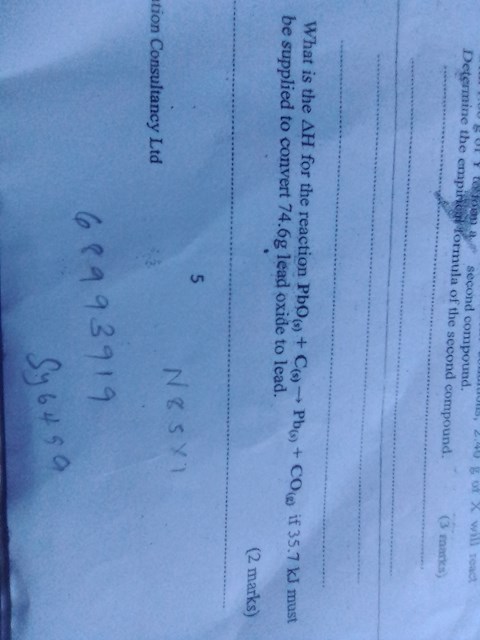#thermodynamics
Thermodynamics - questions and answers. If you have a question about this topic, please click on the "New Question" button. If you wouldn't like to miss any new question in this topic, please subscribe.

Simon Petergeminia
Apr 25

Simon Petergeminia
Apr 25

Simon Petergeminia
Apr 25
Kiconco Analena
Apr 15

Qedani Mkhonza
Apr 12
Anonymous
Apr 8
Boiling point increases as molar heat of vaporization increases however it will not determine the strength of the intermolecular forces between molecules. true or false?
Boiling point increases as molar heat of vaporization increases however it will not determine the strength of the intermolecular forces between molecules. true or false?
1
1
Anonymous
Apr 8

Derek Moenga
Mar 31

Derek Moenga
Mar 31
Anonymous
Mar 31

Derek Moenga
Mar 31